I. Introduction
1.1 Research objectives and questions
II. Methods
2.1 Identifying the research question
2.2 Identifying relevant studies
2.3 Study selection
2.4 Charting the data
2.5 Collating, summarizing, and reporting the results
III. Results
3.1 General characteristics of soundbased multisensory interventions
3.2 Intervention physical environments and temporal characteristics of sound based multisensory interventions
3.3 Types and characteristics of sensory stimuli of sound based multisensory interventions
3.4 Functional roles of sound in soundbased multisensory interventions
IV. Discussion
4.1 General characteristics
4.2 Types, characteristics, and technical platforms
4.3 Role and mechanism of sound
V. Conclusions
Appendix A. Supplementary material
I. Introduction
Sleep disorders are prevalent and affect millions of people worldwide. In the United States of America, more than one-third of the population experiences insufficient sleep, and has a prevalence of short sleep duration (≤ 6 h) among adults, which increased from 22.3 % in 1985 to 32.9 % in 2017,[1,2,3] In Europe, approximately 45 million people have by sleep disorders.[4]
One of the most common sleep disorders is insomnia.[5] Insomnia is characterized by difficulty in initiating or maintaining sleep and waking up too early at least three times per week over a period of 3 months.[6,7,8] Populationbased research indicates that around 30 % of adults report insomnia symptoms annually, with 60 % ~ 10 % meeting chronic criteria.[9,10,11]
Insomnia is associated with marked functional impairment and comorbidities, including obesity, diabetes, cardiovascular diseases, and mental health disorders such as depression and anxiety.[12,13,14] The economic burden of insomnia is substantial, with annual losses in the United States of America estimated at $100 billion USD.[15]
Pharmacotherapy is one of the primary methods for treating acute insomnia. However, pharmacotherapy can have limited efficacy, potential for dependency, and adverse effects.[16] Cognitive Behavioral Therapy for Insomnia (CBT-I) is among the most prevalent nonpharmacological interventions.[17] CBT-I faces practical challenges, including limited accessibility and high cost.[18] Despite these limitations, numerous studies have demonstrated its effectiveness.[19,20,21] International guidelines recommend the combined use of pharmacotherapy and CBT-I as an effective insomnia treatment.
Auditory stimuli is a promising approach for promoting sleep.[22] Insomnia is considered to be caused by abnormal neuronal activity in areas such as the thalamus, frontal lobe, parietal lobe, brainstem, cerebellum, and caudate nucleus.[23] Auditory stimulation can modulate neural activity in these regions by inducing changes in relative power and brain connectivity in specific frequency bands, which may contribute to the regulation of neural activity associated with sleep.[24] Auditory stimulation increases the electroencephalogram (EEG) power by targeting slow waves (delta waves) during deep sleep[25] and supports encoding activation in the hippocampus, which facilitates functional activity during sleep.[26] It also increases the parasympathetic nerve activity, which promotes sleep recovery.[27] Auditory stimuli include music, white noise, pink noise, and binaural beats. All of these have been proven to be effective as adjunctive or preventive sleep interventions.[28,29,30,31] These auditory stimuli can synchronize brain waves to facilitate desired sleep states[32,33] or block out unwanted noise to enhance psychological relaxation and promote sleep.[34]
In addition to auditory stimuli, visual, tactile, and olfactory stimuli have also been used for insomnia interventions. Visual stimuli, such as photic stimuli, can reduce sedative use and alleviate patient discomfort by triggering slow-wave responses.[35] Studies on the impact of tactile stimuli on sleep have included those on vibrations and massage. Vibrations at low frequencies, also known as beat frequency vibrations, and low-frequency vibrations produced by mechanical beds can promote or facilitate sleep.[36,37] Olfactory stimuli such as aromatherapy also substantially enhance sleep quality. In various populations, including patients with diabetes, middle-aged women with insomnia, and university students, aromatherapy has improved sleep quality.[38,39,40]
These findings demonstrate that individual sensory modalities exert various beneficial effects on sleep. Among these, auditory stimulation is a powerful stimulus that directly modulates brain waves and neural activity.[41] When combined with other sensory stimuli, auditory stimulation can multiply the neurophysiological effects and maximize sleep quality.[42] Therefore, a multisensory approach centered on auditory stimulation that combines a variety of sensory stimuli can be very effective. For example, music can be combined with tactile stimuli to create immersive and emotionally rewarding sensory experience.[43] Similarly, the intensity and pleasantness of olfactory stimuli are enhanced when auditory and olfactory stimuli are combined. Thus, sensory interactions can improve the overall sensory experience,[44,45] which can be used to encourage better sleep. However, despite their potential, criteria for combining various sensory modalities to enhance sleep remain unclear. To resolve this issue, it is necessary to conduct comprehensive evaluations to identify effective mechanisms and their combinations.
This scoping review systematically examined the range and characteristics of sound based multisensory sleep interventions. Preliminary searches indicated that no existing scoping review has addressed this topic, highlighting the need for foundational work in this area. Accordingly, clarifying the scope, methodological features, and sensory configurations of prior studies is essential for understanding how sound interacts with other sensory modalities to influence sleep. By synthesizing prior research on sound based multisensory interventions, this review provides an overview of how auditory stimulation interacts with other sensory modalities and which sensory combinations have been most commonly applied in sleep-related studies. These consolidated findings offer a basis that can inform the design of more evidence based and theoretically refined multisensory sleep interventions and can assist in building methodological approaches for evaluating their effects. Overall, this review serves as a foundational resource that can support subsequent research exploring multisensory approaches to improving sleep.
1.1 Research objectives and questions
The primary objective of this study was to comprehensively review the existing research on sound based multisensory sleep interventions and understand their effects and roles. Therefore the following research questions were established.
1) What are the general characteristics of sound based multisensory interventions?
2) What types, characteristics, and technical platforms are used in sound based multisensory interventions?
3) What are the functional roles and mechanisms of sound in sound based multisensory interventions?
II. Methods
The Joanna Briggs Institute (JBI) methodology for scoping reviews[46] and the expanded Preferred Reporting Items for Systematic Reviews and meta-analyses guidelines for scoping reviews (PRISMA-ScR)[47,48] were used to conduct this scoping review.
2.1 Identifying the research question
The research questions of this study focused on analyzing how sound based multisensory interventions for sleep are defined and utilized. Key details about the authors, publication years, study designs, and participant information, which included the diagnosis, type, primary condition, age, sex, and number of participants with insomnia were obtained from each study. Temporal aspects, including session durations, frequency of interventions, and overall intervention durations were analyzed along with the physical contexts in which they were implemented. The investigation also focused on the varieties and characteristics of the auditory stimuli employed as well as their integration with other sensory inputs, including visual, tactile, and olfactory stimuli. The major outcomes of these therapies were evaluated, with particular emphasis on their effects on sleep quality and other pertinent sleep metrics. The functional roles and mechanisms of sound in various interventions were analyzed to assess the specificity, purpose, and overall efficacy of sound in improving sleep outcomes.
2.2 Identifying relevant studies
2.2.1 Inclusion criteria
Following the JBI methodology, eligibility criteria were set based on population (with no restrictions), concept (including all sound based multisensory interventions), and context (covering various settings related to sleep health and medicine).[49] The inclusion criteria were defined based on these Population-Concept-Context (PCC) elements according to the following review topics
2.2.1.1 Participants
The participants included individuals with sleep problems without sex or age restrictions. Articles on insomnia as a primary condition as well as insomnia resulting from other conditions were included. Articles with participants with comorbidities were also included if sleep difficulties were the primary focus.
2.2.1.2 Concept
This concept was centered on articles that addressed sound based multisensory interventions for sleep. In addition to visual, tactile, and olfactory stimuli, a variety of auditory stimuli, including music, white noise, pink noise, and binaural rhythms were included. These studies also explored how these interventions affected sleep quality and other sleep-related outcomes, even if their primary focus was not insomnia. Simultaneous and sequential stimuli were included to cover a wide range of intervention designs.
2.2.1.3 Context
The context was broad and consisted of various settings related to sleep health and sleep medicine, without limitations on demographic factors or specific environments. This encompasses research conducted in both clinical and non-clinical environments including laboratories, personal environments, nursing homes, and hospitals. The emphasis was on sound based multisensory interventions designed to enhance sleep quality and alleviate insomnia, irrespective of the specific environment in which the studies were conducted.
2.2.2 Exclusion criteria
Full-text articles were excluded if they were inaccessible. Furthermore, articles that did not correspond to the objectives of this review were excluded because of incorrect research design. Duplicate publications, including those incorporated into systematic reviews, were also excluded. In addition, conference abstracts, book sections, and articles that were not published in English were excluded.
2.2.3 Search strategy
This study utilized three online databases, Scopus, Web of Science, and PubMed, to identify relevant literature. The topic was extensively investigated, and the search was expanded by utilizing a combination of keywords and Boolean operators “AND” and “OR.” We conducted a search for articles published between January 1, 2014, and April 11, 2024. The search was conducted using the following keywords: “acoustic” OR “auditory” OR “audio*” OR “music*” OR “sound” OR “noise” AND “visuo” OR “visual” OR “tactile” OR “vibr*” OR “haptic” OR “touch” OR “olfact*” OR “odor” OR “aroma*” OR “fragran*” OR “scent” AND “intervention” OR “treatment” OR “stimulation” OR “therap*” OR “protocol” OR “induc*” OR “improve*” OR “enhanc*” AND “sleep*” OR “nap” OR “insomnia”
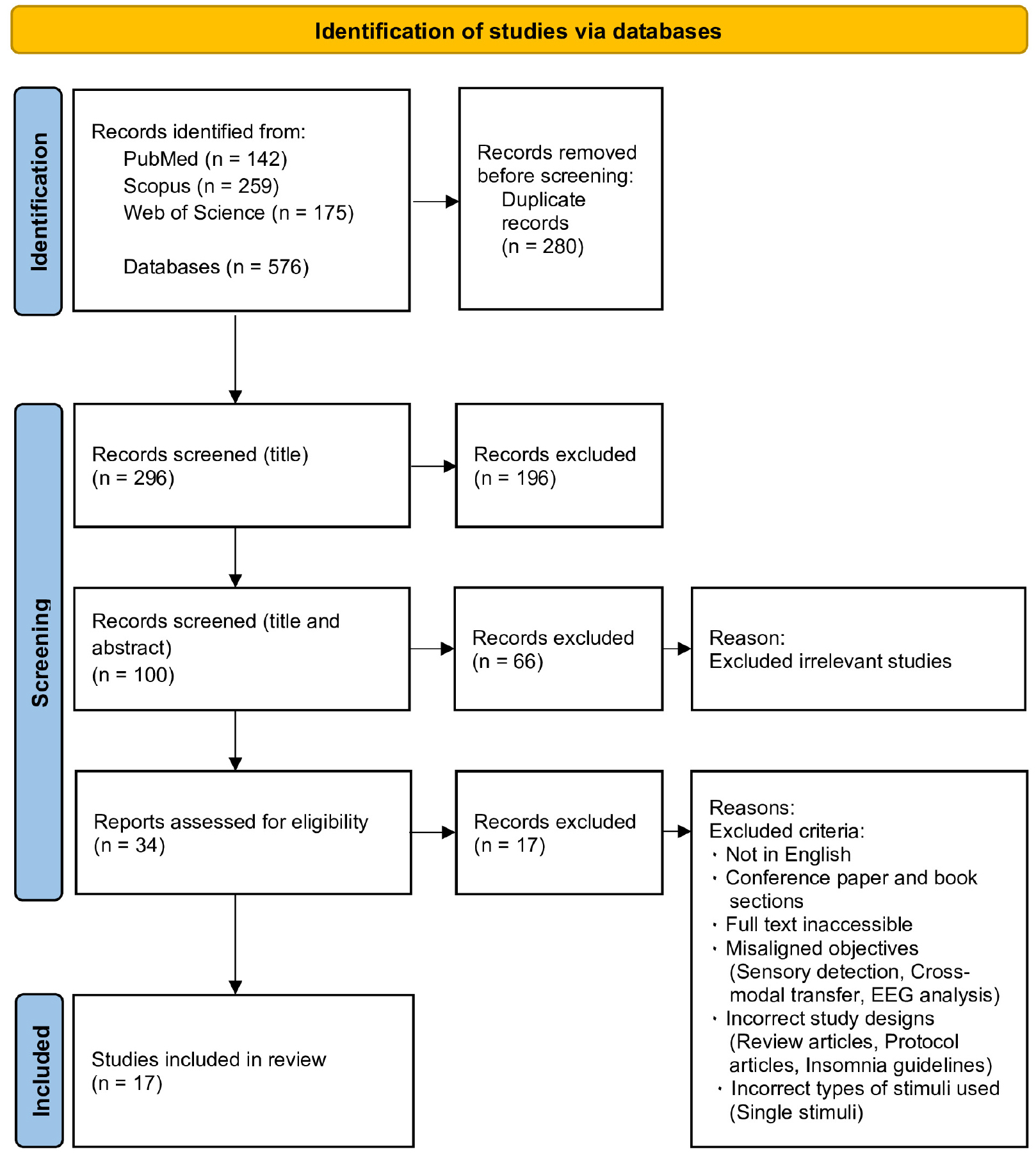
After the search, all identified citations were collated and uploaded into Zotero 6.0.36 (Roy Rosenzweig Center for History and New Media, George Mason University, Fairfax, VA, USA), and duplicates were removed. Two independent reviewers conducted a pilot test and screened titles and abstracts for eligibility based on the inclusion criteria. The full texts of the potentially relevant sources were subsequently evaluated in depth by the same two reviewers. Exclusions of full-text sources that failed to meet the inclusion criteria were documented and presented in the scoping review. Discrepancies among the reviewers at each phase of the selection process were addressed through discussions or by consulting a third reviewer. The outcomes of the search and study inclusion procedures were comprehensively detailed in the final scoping review and presented in a PRISMA-ScR flow diagram (Fig. 1).
2.3 Study selection
Articles from the three databases were identified, checked for duplication, and screened. The initial search yielded 576 articles. After removing 280 duplicate studies, 296 were identified. The screening process comprised three stages: title screening, title and abstract screening, and full-text screening. Based on the inclusion and exclusion criteria described in subsection 2.2, 100 articles were selected for title screening and 196 articles were excluded. Of the 100 articles, 66 were excluded during the title and abstract screening phases because they did not meet the language criteria, had incorrect study designs (review articles or insomnia guidelines), were conference abstracts, or book sections. Finally, 34 articles were selected for further screening. After full-text screening, 17 articles were selected for inclusion. The articles excluded at this stage either did not meet the aforementioned language criteria, had incorrect study designs, used incorrect types of stimuli (such as single stimuli), or had research objectives that were not aligned with this study, such as sensory detection, cross-modal transfer, and EEG analysis. The literature search and selection processes are displayed in Fig. 1.
2.4 Charting the data
To ensure systematic collection and organization of data for this study, we reviewed 17 carefully selected articles. General information, physical environments, temporal characteristics, types and characteristics of interventions, and the functional role of sound were compiled for each article. During data entry, two researchers independently extracted and cross-verified the data to improve accuracy and validity.
2.5 Collating, summarizing, and reporting the results
This study used a descriptive summary and thematic analysis, and employed data from each article. The descriptive summary comprised a numerical analysis of the general information presented in the publications, whereas the thematic analysis aimed to identify significant themes related to the research aims and investigated their prevalence across numerous studies. Through close integration of these themes with the data, we intended to gain a clear understanding of the significance and outcomes of the research. The framework for the data analysis is presented in Table 1.
Table 1.
Framework for the data analysis.
The analysis entailed the collection and categorization of data according to the descriptive information provided by the researchers. A thematic analysis of the sound information was subsequently performed, with themes classified and analyzed based on their functional roles. This technique involved identifying critical keywords and core contents from each study and categorizing them based on their significance and context.
III. Results
3.1 General characteristics of soundbased multisensory interventions
In this study, 17 articles were selected for final analyses. Table 2 summarizes the details of each article and provides a comprehensive overview, divided into two categories: study identification and participant details. The study identification section includes the authors, publication years, and study designs for each article. The participant details section includes the insomnia type diagnosed and age classifications. Detailed study identification and participant characteristics including authors, participant primary conditions, mean age, sex, and sample size, are available in the supplementary material Tables A1.
Table 2.
Distribution of general characteristics for the included studies.
3.1.1 Study identification
None of the selected articles were published in 2016 or 2018. The year 2022 had the highest number of publications, with four papers (23.5 %) (Table 3). Table 3 illustrates that the study designs were diverse, with Randomized Controlled Trials (RCTs) being the most prevalent (29.4 %), followed by quasi-experimental studies (23.5 %), Pre-Post experimental designs (Pre-Post) (17.6 %), randomized pre-post designs (11.8 %), randomized crossover trials (11.8 %), and single-blind RCTs (5.9 %).
Table 3.
Characteristics of intervention environments and durations for the included studies.
3.1.2 Participant details
The participants were classified based on their insomnia diagnosis and type. They were categorized into three groups: those officially diagnosed with insomnia, those exhibiting symptoms and without an official diagnosis, and those without insomnia symptoms. Studies involving participants with no insomnia symptoms were the most common (52.9 %), followed by those with symptoms but no official diagnosis (35.3 %), and those with an official diagnosis of insomnia (11.8 %) (Table 3). Insomnia severity was primarily measured using the Insomnia Severity Index (ISI). All participants who were officially diagnosed with insomnia were classified as having chronic insomnia, which accounted for two of the studies. The primary conditions of the participants included a wide range of disorders such as general insomnia, autism spectrum disorder, coronary artery disease, chronic low back pain, congenital abdominal disorders, moderate burns, osteoarthritis, and Alzheimer’s disease. The participant age categories were diverse: adults aged 18 to 49 years (52.9 %), older adults aged 65 years and older (17.6 %), adults aged 50 to 64 years (11.8 %), children and adolescents (11.8 %), and neonates (5.9 %) (Table 3). The sex distribution indicated that females (65.6 %) were more frequently included than males (34.4 %).
3.2 Intervention physical environments and temporal characteristics of sound based multisensory interventions
3.2.1 Intervention environments
The intervention environments in this study varied across numerous research contexts, and comprised home settings, hospitals, research institutes, laboratories, and sanatoriums. Environmental selection was determined by the participants and aims of each study. Home settings were most common (47.1 %), followed by hospitals (29.4 %), research institutes (5.9 %), combinations of research institutes and home settings (5.9 %), laboratories (5.9 %), and sanatoriums (5.9 %). The variations in the intervention environments are summarized in Table 3.
3.2.2 Intervention duration and frequency
The duration and frequency of interventions varied significantly. Each session ranged from a few minutes to several hours, and the overall intervention period varied from 1 day to 8 months. Interventions were conducted once daily to twice weekly. The variations in duration and frequency are summarized in Table 3.
3.3 Types and characteristics of sensory stimuli of sound based multisensory interventions
In the included studies, sound based multisensory interventions combined auditory stimuli with visual, tactile, and olfactory inputs. For the purposes of this review, these interventions were grouped into five sensory combination types: (1) auditory-visual, (2) auditory-tactile, (3) auditory-tactile-olfactory, (4) auditory-olfactory, and (5) auditory-visual-tactile (haptic). In this review, the term tactile is used as an umbrella concept referring to skin-mediated sensory input, including vibration, pressure, massage, and gentle touch. One included study implemented an auditory-visual-tactile configuration in which the tactile component was an acupressure based wrist cuff device that the authors explicitly designated as haptic in the original publication. To maintain clarity and avoid unnecessary terminology overlap, we conceptually situate this haptic stimulation within the broader tactile domain, while retaining the original term haptic when referring to that specific intervention as reported by the authors.
3.3.1 Frequency of sensory combinations and methods of presenting sensory stimuli
The most common multisensory approaches selected were auditory-visual combinations (41.2 %), which included methods such as flickering lights with pulsing and binaural sounds or videos paired with music and Light Emitting Diode (LED) lights with noise. Auditory-tactile combinations (29.4 %) included interventions such as massage, therapeutic touch, and vibrations, all of which were paired with music. The auditory-tactile-olfactory combinations (17.6 %) included aromatherapy and massage accompanied by music. Combinations of auditory-visual-tactile (haptic) (5.9 %) and auditory-olfactory (5.9 %) modalities were infrequent and utilized mixed sensory inputs such as LED lights paired with binaural sounds and wrist cuff pressure, and aroma oil combined with nature sounds (Table 4).
Table 4.
Sensory combination types, characteristics, and presentation methods of sensory stimuli in the included studies.
Simultaneous methods were implemented in all investigations (100 %) to present multiple sensory inputs concurrently. Although Zabrecky et al.[50] conducted a study in which auditory and vibratory stimuli were administered simultaneously with additional auditory stimuli delivered sequentially, the simultaneous presentation of stimuli remained the predominant method across all studies (Table 4).
3.3.2 Detailed profiles of sensory stimuli combinations
Each sensory combination exhibited distinct characteristics in the way stimuli were presented. Auditory-visual stimuli were applied extensively in research to synchronize brainwave activity with sleep-related EEG patterns, particularly for guiding the brain toward delta wave activity. For example, the delivery of pulsing sounds and flashing lights resulted in a gradual decrease in stimulation frequencies; the objective of this combination was to increase delta brainwave activity.[51,52,53] Another approach involved the use of a personalized sleep mask that integrated underwater marine videos, audio, and biofeedback.[54] A single study employed red LED light in conjunction with pink noise stimuli to induce a more profound sleep state.[55] Additionally, 40 Hz gamma auditory-visual stimulation was used in certain studies to enhance gamma brainwave activity, which is associated with cognitive functions, and explored in the context of improving sleep quality for patients with Alzheimer’s disease[56]. Additionally, popular Autonomous Sensory Meridian Response (ASMR) content that incorporates auditory stimuli and visual components has been implemented.[57]
The auditory-tactile combinations included massage and soothing touch with music, low-frequency vibration stimuli paired with low-frequency sine wave sounds, and vibrations synchronized with music. For example, massage and therapeutic touch were administered prior to, during, and following medical procedures while background music was played.[58,59,60] In addition, low-frequency sine wave auditory and vibration stimuli were implemented to introduce theta and delta waves,[50] and music-mapped vibrations enabled the participants to perceive sound as a tactile sensation.[45]
The auditory-olfactory combinations involved aromatherapy with essential oils while simultaneously listening to nature sounds. For example, to create a relaxing environment, lavender essential oil was applied while sounds of flowing water sounds were played.[61] The auditory-tactile-olfactory combinations included listening to music or nature sounds while receiving an aroma massage applied to the hands, shoulders, feet, and legs using oils such as chamomile, lavender, jojoba, and almond infused with lemon and lavender.[62,63,64] The auditory-visual-tactile (haptic) combinations consisted of stereo or binaural sounds with tones such as 60 Hz and 64 Hz, where the difference between the tones was used to generate a 4 Hz beat, accompanied by red LED lights and wrist cuff pressure.[65]
3.3.3 Technical platforms used for soundbased multisensory interventions
This review included studies that used a variety of technological tools to provide multisensory stimulation to make interventions more precise and effective. Platforms comprised wearable devices, vibro-acoustic systems, automated multisensory stimulation systems, closed-loop neurofeedback systems, and gamma oscillation stimulation systems.
3.3.3.1 Wearable devices systems
Wearable devices are instrumental for consistent and precise delivery of sensory stimulation during sleep. For example, a personal sleep mask that incorporates auditory and visual stimulation can be monitored and altered in real-time according to a user’s vital signs. The mask uses video eyewear to provide high-resolution visual and auditory stimuli and detects breathing patterns via a smartphone placed on the user’s abdomen and adjusts the audio and visual environments in real-time to match slow diaphragmatic breathing. The device induces physiological relaxation by providing an optimal audiovisual experience when the breathing rate reaches a target cycle and by implementing a real-time feedback loop that automatically modifies stimuli based on vital signs.[54] Another wearable device employs technology that gradually lowers the frequency of light flashes and sounds to induce delta-wave activity, which is necessary for sleep promotion. This in turn results in a state of profound relaxation and sleep onset. These audiovisual stimuli enhance the quality of sleep and facilitate the rapid progression to deeper sleep phases by modulating brain waves during sleep initiation and maintenance.[51,52,53]
3.3.3.2 Vibroacoustic systems
Vibroacoustic (VA) stimulation systems deliver sinusoidal noise and vibrations at frequencies ranging from 8 Hz to 10 Hz. These systems are integrated into recliners and are specifically designed to improve sleep quality by allowing users to experience both sound and vibration.[50] Moreover, a sound-to-sleep device converts the sound input into tactile vibrations using a resonator-equipped mattress. This enables users to control the levels of both auditory and tactile stimulations, thereby improving sleep and calming the nervous system. The system was proven effective in a study of children with autism; the combination of sensory input helps reduce sensory processing issues and induce relaxation during sleep.[45]
3.3.3.3 Automated multisensory stimulation system
An automated multisensory stimulation system was designed to facilitate brainwave entrainment, which is considered to enhance sleep quality by employing a combination of auditory, visual, and tactile stimuli. To effectively alleviate insomnia and promote relaxation, this fully automated platform delivers synchronized sensory inputs for three distinct auditory, visual, and tactile stimuli. The system simultaneously supplies binaural beats, LED lights, and pressure stimulation for a predetermined duration initiated by pressing a single button. This method facilitates a physiological response that induces a calming effect and promotes relaxation, thereby improving sleep quality.[65]
3.3.3.4 Closed-loop neurofeedback systems
The closed-loop neurofeedback system is intended to improve sleep quality by delivering real-time sensory stimulation based on EEG data. This system continuously monitors user EEG activity with a focus on slow-wave oscillations, which are critical for restorative sleep. The system dynamically adapts to the user physiological state to facilitate deeper, uninterrupted sleep by synchronizing sensory information with brainwave patterns. This closed-loop feedback system serves as an effective instrument for improving sleep quality owing to the optimization of stimulus timing.[55]
3.3.3.5 Gamma oscillation stimulation system
The gamma oscillation stimulation system employs a combination of light and sound frequencies to synchronize gamma brainwave activity and enhance cognitive performance and sleep quality. The system provides auditory stimulation at 40 Hz, which is linked to gamma brainwave activity, and uses synchronized light at 40 Hz to support brainwave entrainment. It includes a handheld controller for modifying settings, a sleep mask for light stimulation, and headphones for auditory input to provide a customizable experience based on user needs. Furthermore, an actigraphy device tracks sleep patterns by monitoring the movement and rest cycles. Intervention efficacy is assessed by uploading data collected from these devices. Gamma oscillation stimulation systems have been particularly effective for individuals who experience cognitive decline, such for patients with Alzheimer’s disease. By promoting gamma brainwave activity, the system not only enhances cognitive function but also improves sleep quality.[56]
3.4 Functional roles of sound in soundbased multisensory interventions
The synthesis of the reviewed studies allowed the functional roles of sound to be analyzed and categorized. The findings demonstrated that sound served a multitude of functions including synchronizing brainwaves, promoting calm and relaxation, managing emotions and stress, dual purposes of noise masking and relaxation, and addressing sensory preferences. Some studies concentrated on the effects of sound in isolation, whereas others explored its role in multisensory stimuli. Consequently, the functional role of sound was categorized as either a unisensory stimulus or a component of a multisensory stimulus, as listed in Table 5.
Table 5.
Distribution of functional roles of sound in the included studies.
3.4.1 Synchronization of brainwaves
Numerous studies demonstrated the significance of sound in modulating brain activity and facilitating sleep. Auditory stimulation affects brainwave patterns at specific frequencies by aligning neural oscillations to enhance sleep onset and duration. Tang et al.[51,52] employed rhythmic pulsing sounds to stimulate sensory pathways, resulting in enhanced neural synchronization and modulation of brainwave activity. Zabrecky et al.[50] utilized sine wave audio to strengthen delta wave patterns, which are essential for sleep, as evidenced by EEG data. Karuppathal et al.[65] and Tang et al.[53] used binaural beats to synchronize brainwaves in the delta range to facilitate deep sleep. Danilenko et al.[55] applied pink noise to enhance power density in the slow-wave band while diminishing it in the rapid spindle band, thereby augmenting restorative sleep quality. Cimenser et al.[56] further investigated the use of 40 Hz auditory stimulation to enhance gamma brainwave activity and the utilization of sine wave audio, which included theta and delta waves, to regulate and improve delta brainwave patterns.
3.4.2 Calming and relaxing effects
Various studies have employed sounds to reduce arousal and improve relaxation. Music was combined with massage, aromatherapy, therapeutic touch, video content, and ASMR videos to achieve a calming effect. James et al. [58] used background music during healing touch therapy to ensure comfort and reduce stress. Smejka and Wiggs[57] employed ASMR videos that included auditory stimulation such as tapping and whispering to promote relaxation and improve mood. Furthermore, Thepsatitporn et al.[61] combined nature sounds, such as flowing water, with aromatherapy to activate the parasympathetic nervous system, which resulted in increased calm and relaxation. de Zambotti et al.[54] used a classical music soundtrack with video content and biofeedback equipment, which allowed participants to alleviate their anxiety and achieve cognitive rest.
3.4.3 Emotional and stress regulation
Several studies emphasized the significance of sound in promoting relaxation and managing stress, both of which contribute to improved emotional and physical health. Choi[62] found that anxiety was significantly reduced with a combination of hand massage and playing preferred songs. Gao et al.[60] used comforting background music to reduce infant anxiety and enhance emotional regulation and relaxation responses immediately before and after medical procedures. Kabuk et al.[59] employed a Huseyni melody along with foot Reflexology Massage (RM) to evoke feelings of relaxation, serenity, and calmness; this combination facilitated the activation of natural healing processes and the restoration of energy balance.
3.4.4 Dual function (noise masking and relaxation)
Sayed et al.[63] demonstrated that combining shoulder aroma massage with slow rhythm and repetitive tempo music not only reduced stress factors but also improved noise levels. Similarly, Nicola et al.[64] discovered that a combination of music, nature sounds, and aromatherapy massage facilitated both relaxation and environmental noise reduction.
3.4.5 Additional functional roles (sensory preferences)
Frazier et al.[42] used customized audio that reflected participant sensory preferences to facilitate sleep onset in children with autism spectrum disorder. The children were permitted to preselect the calming sounds provided or select their own preferred sounds, with the volume adjusted to suit their preferences. This method was designed to create an environment in which children felt comfortable and aided their sleep onset.
IV. Discussion
This review aimed to establish a foundational understanding of how auditory stimuli, often combined with visual, tactile, and olfactory elements, can influence sleep outcomes, and inform future research and practice for sleep enhancement interventions. This scoping review screened 17 studies using the JBI and PRISMA-ScR methodologies. The results highlight that sound based multisensory interventions were versatile, and applicable in clinical and non-clinical populations and settings. The functions and mechanisms of sound based multisensory interventions primarily involved brainwave synchronization, relaxation, and emotional regulation, each of which contributed to enhanced sleep quality.
4.1 General characteristics
The included studies employed diverse designs, with RCTs being most common, followed by quasi-experimental and pre-post experimental designs. Participants were mainly adults (18–49 years, 52.9 %) and older adults (≥65 years, 17.6 %), with women comprising 65.6 %. Trials were typically conducted at home (47.1 %), though hospitals, research institutes, and sanatoria were also used. Intervention durations ranged from one day to several months.
Our review primarily focused on adult populations, whereas other studies included older adults, children, adolescents, and neonates. Given that the causes and symptoms of insomnia vary significantly across age groups, tailored approaches and interventions are often required.[66,67,68] Future research should include a balanced representation of various age groups. This would allow for a more comprehensive understanding of how intervention effectiveness differs across age groups.
The majority of studies were conducted in home settings, followed by hospitals and other controlled environments. Home settings provide a realistic sleep environment that closely reflects real-world conditions; however, they also introduce uncontrollable external factors that can influence results. In contrast, controlled environments offer better consistency although they may not accurately represent participants’ typical sleep conditions.[69,70,71] To ensure consistency in findings, future studies should compare outcomes from controlled laboratory settings with those from natural sleep environments.
The duration and frequency of interventions varied across trials, ranging from one day to several months. Although these variations are often tailored to the specific goals of each study, they may hinder the comparability of results due to inconsistent data collection regarding intervention length and frequency. Future studies should adopt standardized protocols to establish consistent intervention frequency, session duration, and total duration, enabling more reliable comparisons and interpretations of the findings.[72,73]
4.2 Types, characteristics, and technical platforms
Multisensory combinations have been classified as auditory-visual, auditory-tactile, auditory-olfactory, auditory-tactile-olfactory, and auditory-visual-tactile (haptic). Auditory-visual stimuli were used most often, followed by auditory-tactile, auditory-tactile-olfactory, and auditory-olfactory and auditory-visual-tactile (haptic) combinations. The majority of studies employed the simultaneous delivery of multisensory inputs, except one study that used a combination of periodic sequential and simultaneous inputs.
Auditory stimuli induce brainwave synchronization or facilitate relaxation based on frequency bands and emotional qualities. Binaural beats increase delta waves by presenting different frequencies to each ear. Music stimulation extends beyond frequency effects by incorporating auditory features that provide emotional relaxation and stability, thereby alleviating stress and tension. The acoustic qualities of music, such as a slow and steady tempo, soft volume, and consistent texture, promote psychological calm and support sleep preparation.[74]
Visual stimuli were used to synchronize brainwaves, mainly with flickering lights or LEDs, and were complementary when combined with auditory inputs. For example, flickering light with pulsing sounds has been used to induce delta waves, while visual and auditory stimulation at 40 Hz activated gamma waves. Visual stimuli also included nature based video content combined with music or natural sounds to create calming experiences.
Tactile stimulation was delivered in the form of low-frequency vibrations synchronized with music, or massages combined with musical elements. Low-frequency vibrations were synchronized with music to alleviate physical tension, whereas massages were designed to promote both physical relaxation and emotional well-being.
Olfactory stimulation was applied as aromatherapy with essential oils combined with nature sounds. For example, lavender scent with flowing water sounds was used to promote relaxation and support sleep.
Consequently, multisensory stimulation compensates for individual sensory stimuli limitations and provides complementary effects that activate multiple sensory pathways.[61] Further research is required to clearly understand the differential mechanisms of this combined approach and the interaction between each sensory stimulus.
Various technological devices and systems have been used to deliver multisensory interventions including wearable devices, vibroacoustic systems, closed-loop neurofeedback systems, gamma oscillation stimulation systems, and automated multisensory stimulation systems. Each offers distinctive features specifically designed to engage a particular combination of sensory modalities.
Auditory-visual combinations were mainly implemented with wearable devices and gamma-oscillation stimulation systems. Wearables such as headbands and sleep masks synchronize auditory and visual inputs using LEDs with pulsed sounds or binaural beats to facilitate brainwave synchronization.[51,52,53] Sleep masks can provide customized multisensory experiences by combining nature based visual scenes with music, while physiological sensors adjust stimulus intensity and frequency according to the user’s state.[54] Gamma-oscillation systems activate gamma waves by synchronizing auditory tones and LED flashes at 40 Hz, ensuring coherence of sensory inputs, and have also been applied in neurocognitive interventions such as Alzheimer’s disease.[56] Closed-loop neurofeedback systems dynamically adjust auditory and visual stimuli based on real-time EEG monitoring to enhance slow-wave sleep. This feedback mechanism ensures that sensory inputs are precisely aligned with brain activity, amplifying slow-wave oscillations and improving sleep quality in a personalized and adaptive manner[55].
The auditory-tactile combination was implemented using two distinct vibroacoustic systems. The first system, integrated into a recliner chair, delivered synchronized auditory and vibration stimuli designed to induce physical relaxation through alignment with theta-wave frequencies.[50] The second system, a Sound-to-Sleep mattress (STS), generated sound and vibration simultaneously by converting musical rhythm and frequency into physical vibrations, integrated auditory and tactile stimulation to enhance the user’s sensory experience. This technology has been used to relieve stress and support sleep preparation.[45]
The auditory-visual-tactile (haptic) combination was supported by an automated multisensory stimulation system. This system delivers binaural beats, red LED lights, and HT-7 acupressure stimulation in a fully automated and synchronized manner. This system simultaneously activated all three modalities to align brainwaves, promote melatonin production, and reduce stress.
Auditory-olfactory and auditory-olfactory-tactile combinations relied on traditional methods rather than advanced technology platforms, typically combining music or nature sounds with aromatherapy. Examples include lavender scent with water sounds or soft music, and aromatherapy massage with relaxing melodies to create multisensory experiences.[61,62,63,64] Unlike other combinations, these approaches did not employ specialized technology platforms.
The technological platforms and methods used to deliver various sensory stimuli highlighted the applicability of multisensory combinations. Advanced systems such as wearable devices, gamma oscillation stimulation, closed-loop neurofeedback, vibroacoustic systems, and automated stimulation were primarily applied to auditory-visual, auditory-tactile, and auditory-visual-tactile combinations, where precise synchronization of stimuli is critical. These platforms demonstrated their technological capacity to enhance sensory experiences through frequency tuning, real-time adjustment, and seamless multimodal delivery. In contrast, traditional approaches, such as combining music or nature sounds with aromatherapy or massage, were predominantly applied to auditory-olfactory and auditory-olfactory-tactile combinations, relying more heavily on natural sensory inputs. Despite these findings, research on how each sensory combination can be technologically optimized to maximize effectiveness remains limited. Future research should focus on determining the most effective combinations and delivery methods using specific platforms.
4.3 Role and mechanism of sound
The functional roles of sound in sleep interventions can be categorized as brainwave synchronization, calming and relaxing effects, emotional and stress regulation, dual functions (relaxation and noise masking), and sensory preferences. Binaural rhythms and other auditory stimuli synchronize EEG activity with sleep-promoting frequencies such as delta and theta waves, facilitating deeper sleep.[50,51,52,53,55,56,65] Beyond brainwave synchronization, sound is essential for promoting calming and relaxing effects and is usually combined with tactile cues (massage), olfactory cues (fragrance), and visual cues (videos) to lower physiological arousal and stress.[55,57,61,66] Music is particularly effective in promoting autonomic nervous system restoration, modulating cortisol responses, and evoking feelings of comfort and stability to promote relaxation and calm.[75] This relaxation effect induced by sound based interventions improves overall sleep efficiency by directly correlating with shorter sleep latency and earlier transition to deeper sleep stages.[76,77] These findings align with the effects observed in multisensory combinations, in which auditory stimuli play a critical role in fostering relaxation and stress reduction. Achieving a state of relaxation is recognized as a fundamental process for sleep.[78,79]
Furthermore, sound plays a key role in emotional and stress regulation by alleviating tension and anxiety, both of which impair sleep quality, thereby creating a conducive environment for sleep onset.[59,60,62] Music modulates the activity in brain regions involved in emotional processing, such as the amygdala and prefrontal cortex, and can contribute to reducing negative emotions and stress.[80,81,82] The effects of music on emotional regulation go beyond mere emotional relaxation and include physiological effects through modulation of the autonomic nervous system, which contributes to stress reduction. These physiological responses lead to improved sleep efficiency and emotional states, and are closely linked to emotional stabilization effects, particularly anxiety reduction.[83,84] Sound has the potential to improve mental health beyond physical relaxation, especially in individuals with insomnia caused by stress or anxiety. Stress and anxiety are major causes of sleep disturbance,[85,86,87] and reducing worry before bedtime is strongly associated with shortened sleep latency and improved sleep stage continuity.[88,89]
In addition, sound performs the dual functions of masking external noise and inducing relaxation. The noise-masking effect minimizes disruptions from environmental sounds, such as alarms or conversations, whereas relaxation-inducing properties help lower physiological arousal. This dual functionality creates a calmer sleep environment, simultaneously reduces external and internal stressors, and significantly enhances the efficacy of interventions by enabling more stable and restorative sleep patterns.[63,64]
Finally, aligning auditory stimuli with participant sensory preferences significantly enhanced the effectiveness of sound based interventions. This was evident for individuals with distinct auditory modality preferences and tailoring sounds to their preferred frequencies, tones, or rhythms ensured better engagement and responses. Such customization not only increases intervention efficacy but also fosters a sense of comfort and acceptance, creating a conducive environment for improved sleep outcomes.[45] These findings underscore the importance of adopting a personalized approach in designing sound based interventions to optimize their impact on sleep quality and overall well-being.
V. Conclusions
This scoping review synthesized existing research on sound based multisensory sleep interventions by identifying the sensory combinations used, the characteristics of each sensory stimulus, and the functional roles of sound. Across the included studies, auditory–visual combinations supported brainwave synchronization to facilitate transitions into deeper stages of sleep; auditory–tactile combinations enhanced physical relaxation and psychological calmness to improve sleep efficiency; and auditory-olfactory combinations contributed to improved sleep quality by creating a more emotionally comforting sleep environment. Taken together, these findings indicate that combining auditory stimulation with other sensory modalities may influence multiple dimensions of sleep.
Despite these observations, important gaps remain. Although the functional roles of sound were categorized into brainwave synchronization, calming and relaxing effects, emotional and stress regulation, and dual functions (noise masking and relaxation), the underlying neurophysiological and emotional mechanisms through which these effects occur are not yet clearly understood. Many studies used broad or nonspecific descriptors for sound stimuli, such as “soft background music,” without reporting key acoustic properties. Furthermore, few studies systematically compared sensory combinations or examined how specific sound characteristics affect sleep-related outcomes.
Future research should systematically compare differences across sensory combinations and analyze how specific sound characteristics, including frequency bands, intensity, and musical style, influence sleep-related indicators. Although existing studies have employed EEG analyses, autonomic nervous system indices, and validated subjective scales, these measures have been applied inconsistently across studies. Therefore, future work should use these indicators within a more standardized and integrated assessment framework to better link subjective sleep experiences with objective physiological changes. Such efforts will help clarify the neurophysiological and emotional mechanisms through which sound based multisensory stimulation influences sleep and support the development of more theoretically grounded and tailored multisensory sleep interventions. Overall, this review provides an evidence-informed foundation for the design and evaluation of sound centered multisensory sleep interventions and serves as a key resource that supports future research on multisensory approaches to improving sleep.