I. Introduction
Cyclodextrins (CDs) (host) are cyclic oligosac-charides composed of glucopyranose units linked by an α‐(1→4) glucosidic bond. Three types of CDs naturally occur with 6, 7, and 8 glucopyranose units, usually referred to as α‐, β‐, and γ‐CD [1]. They are doughnut-shaped molecules that have hydrophobic cavities that can form inclusion complexes with various kinds of molecules. Accordingly, cyclodextrins have been widely used in various fields, such as a drug delivery system, a separation system, and a model for enzymatic specificity. An origin for such interesting applicabilities aries from a specific inclusion ability of cyclodextrins (hosts) for various compounds (guests). A lot of equilibrium studies have been carried out using UV, NMR, titration calorimetry, and so on in order to determine the stability of complexes formed by hosts and guests [2-5].
Kinetic information plays an important role for understanding more precisely why the stability of the complexes depends on guest or host structures and how long the included molecules can stay in cyclodextrin cavities.
The ultrasonic relaxation method is useful for probing the microscopic solution structure and can be used to study fast processes with characteristic time constant of 10-6–10-10 sec. Dynamic association and dissociation processes between host and guest are of fundamental importance for certain functions of supra- molecular systems. The dynamic properties of the interaction between cyclodextrins and nonelectrolytes have been examined in our series of experimental study by the ultrasonic relaxation method [6-8]. These investigations have revealed that the stabilization of the complexes is sensitive to the size of cavity, and to the balance between hydrophobicity and the hydrophilicity of the guest. It is interesting and desirable to examine how the presence of charge on guest molecule affects the dynamical characteristics for host-guest complex formation process because the knowledge of these rate constants has direct applications when using cyclodextrins to perform specific functions. To investigate this charge effect in this study, butanoic acid has been selected as the guest for β-cyclodextrin (β-CD), and two different pH conditions were applied. This kinetic information will serve as an important part of the molecular recognition in the host-guest interaction between β-CD and butanoic acid.
II. Methods
β-CD was purchased from Sigma (St.Louis, MO), and was used without further purification. Butanoic acid was also from the same company in their regent grade. The purity of butanoic acid was confirmed to be better than 99 %. Distilled and filtered water was used as a solvent for all solutions after degassing under reduced pressure. The solutions of butanoic acid were prepared by adjusting the pH≈2.5 with a concentrated aqueous solution of HCl. The dissociated butanoic acid solutions at pH≈6.0 were obtained by the addition of concentrated aqueous solution of NaOH. Experiments were carried out at 25℃ with a temperature control within ± 0.1℃. Solution densities were measured by a vibrating density meter (Anton Paar DMA 5000 N). We used three experimental techniques to measure ultrasonic absorption covering a wide frequency range of 0.2-45 MHz: using a plano- concave resonance method (0.2-1.7 MHz) [9], plano- plano resonance method (2.4-7.5 MHz) [9], and pulse method (25-45 MHz) [10]. Sound velocity values were obtained by the pulse technique resonance method at the 3 MHz. Solution pHs were received through a glass electrode (691 Ω Metrohm pH meter) in the same thermostatic bath for the resonance method. The ultrasonic absorption, sound velocity, and density of the solutions have been measured as a function of the solute concentrations.
III. Results
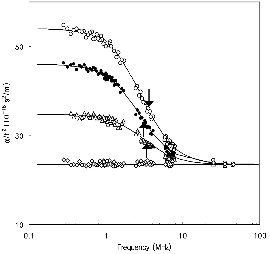
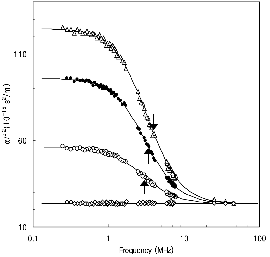
Figures 1 and 2 show representative ultrasonic absorption measurement spectra in aqueous solutions of nonionized butanoic acid (at pH≈2.5) and ionized butanoic acid (at pH≈6.0) in the presence and absence of β-CD, respectively. When β-CD does not exist in the solution (only the guest molecules are dissolved in water), the absorption coefficients divided by the square of the measurement frequency, α/f2 are independent of the frequency: no excess absorption is observed. It is also reported that the ultrasonic relaxation is observed in aqueous solutions of β-CD above 0.013 mol dm-3[11], while the relaxation is not observed below this concentration. The concentration of β-CD was fixed at 0.0087 mol dm-3 in this study. When butanoic acid was dissolved in a solution in the presence of β-CD, the excess absorption was clearly observed. A simple analytical Debye-type relaxation equation [12] is used to make clear the frequency dependence of the absorption
α/f2 = A/{1 + (f/fr)}2 + B (1)
where fr is the relaxation frequency, and A and B are constants. The ultrasonic parameters, fr, A, and B, were determined by fitting the experimental data to equation (1). The α/f2 versus f plot in Figure 1 exhibits a good agreement between the calculated line and the experimental data. This confirms that a single relaxation process is involved in the formation of the inclusion complex. The ultrasonic parameters determined in this study are listed in Table 1 along with the values of the sound velocity, υ, and density, ρ. The magnitude of the background absorption, B, is close to that of solvent water, α/f2=23.5×10-15 m/s2. Since the relaxation appeared only when the two solutes were resolved in water, the cause of the observed relaxation is evidently due to the dynamic interactions between β-CD and butanoic acid. The relaxation from a perturbation of the chemical equilibrium by ultrasonic waves can be described by the following:
kf
β-CD + BA ⇄ β-CDBA (2)
kb
where β-CD is the host, BA is the guest molecule, and β-CDBA is the host-guest inclusion complex. kfand kb are the forward and backward rate constant, respectively. In the analysis of the ultrasonic relaxation, two important quantities should be satisfactorily interpreted. One is the relaxation time, τ, and the other is the amplitude of the relaxation, A. On the basis of a procedure for the chemical relaxation analysis for reaction of equation (2), the relaxation time, τ, is derived as
τ-1 = 2πfr = kf {[β-CD] + [BA]} + kb
= kb{(KCβ-CD + KCBA + 1)2 – 4K2Cβ-CDCBA}1/2
(3)
where K is the equilibrium constant defined as K = kf/kb = [β-CDBA]/[β-CD][BA] and Cβ-CD and CBA are the initial concentrations of the host and the guest, respectively. When Cβ-CD is fixed, fr ought to be dependent only on CBA.
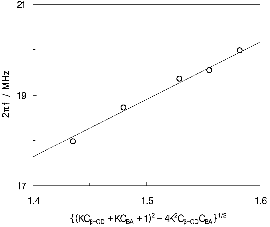
First, the analysis of the results in the nonionized butanoic acid is carried out. Actually, the relaxation frequency increased with an increase of the guest concentration, as can be seen in Table 1. Consequently, the two unknown parameters, K and kb, were estimated by using a nonlinear least-squares method. The results obtained are listed in Table 2. Plot of the 2πfr versus concentration term, {(KCβ-CD + KCBA + 1)2 – 4K2Cβ-CD CBA}1/2 is shown in Figure 3. The solid line in the figure is drawn using the determined K and kb values, and it can be seen that the experimental data are consistent with the calculated line. The good agreement between the calculated line and experimental data supports the validity of the proposed reaction mechanism. The forward rate constant, kf, was obtained from the definition of K, and it is listed in Table 2. A maximum absorption per wavelength, μmax, used for the analysis of the amplitude of the relaxation, is defined as μmax = 0.5Afrυ. This quantity is connected to other thermo-dynamic quantities by the next equation:
|
Fig. 3. Plots of 2πfr vs {(KCβ-CD + KCBA + 1)2 – 4K 2Cβ-CDCBA}1/2 for aqueous solutions of butanoic acid in the presence of 8.7 mM β-CD at 25℃ and pH≈2.5. |
μmax = πρυ2 λ-1 (ΔV)2/2RT (4)
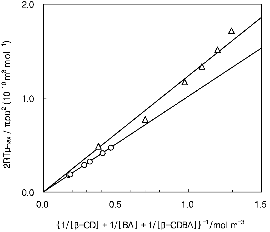
where ΔV is the standard volume change of the reaction, λ is the concentration term as 1/[β-CD] + 1/[BA] + 1/[β-CDBA], R is the gas constant, and T is the absolute temperature. Plots of 2RT μmax / πρυ2 versus λ-1 are shown in Figure 4; from the slope of the lines, the value of ΔV was calculated.
Next, the analysis of the aqueous solution of ionized butanoic acid and β-CD is performed. The relaxation frequencies are independent of CBA, as can be seen in Table 1. However, the relaxational amplitude, A, increased monotonically with CBA, and a similar tendency was also seen when c-AMP , AMP, and isomeric alcohols were the guests for β-CD. The equilibrium concentrations of the reactants in equation (2) can be obtained if the K value is known. Thus, a trial-and-error procedure was used to obtain the K value which provided the best match to the experimental plots of 2RT μmax / πρυ2 versus λ-1, with the line passing through the origin. The best fit is obtained when K= 7.0 dm3 mol-1, which is shown in Figure 4. The ΔV value in Table 2 was calculated from the slope of the line. Using the definition of the equilibrium constant, K, the rearrangement of equation (3) yields the equation for the backward rate constant, kb, as follows:
|
Fig. 4. Plots of 2RTμmax / πρυ2 vs {1/[β-CD] + 1/ [BA] + 1/[β-CDBA]}-1 for aqueous solutions of butanoic acid in the presence of 8.7 mM β-CD at 25℃: (○) pH≈6.0; (△) pH≈2.5. |
kb = 2πfr /[K{[β-CD] + [BA]} +1] (5)
Since the equilibrium constant was determined and the reactant concentrations in equation (5) were available, the value of kb was calculated using the experimental relaxation frequency. The mean value of kb is listed in Table 2. Then, the forward rate constant, kf, were determined with the help of the K values. They are also shown in Table 2.
IV. Discussion
In this study, we demonstrated that the interaction between β-CD and butanoic acid follow a typical spectrum of single relaxation process that results from host-guest interactions. Inclusion complex studies of β-CD with AMP, cAMP, amino acid and isomeric alcohols have shown that the forward rate constant, kf, which is dependent on the diffusion-controlled reaction regardless of guest molecules, is in the order of ~108 M-1s-1. The forward rate constant, kf, of β-CD and butanoic acid obtained in our study is also consistent with the values of the diffusion-controlled reactions [6-8,13]. A comparison of the results of the dissociated form with those in the undissociated form declares that the rate constant for the ionized guest is smaller than that for the neutral guest. The probability for the undissociated guest to encounter the entrance of the cavity is considered to be higher than that for the dissociated guest because the rims of the CD cavity have polar hydroxyl groups.
The dissociation rate constant, kb, is dependent on the structure of the guest molecules. The more hydrophobic the guest molecule is, the slower the dissociation from the host-guest complex is. The similar trends were found in the system with alcohols, AMP, cAMP, and amines as the guests [6-8,13]. These dependences of the rate constants on the hydrophobicity of the guest molecules reflect clearly on the stability of the inclusion complex. Upon increasing the hydro-phobicity of the guest, the complexes are stabilized further, as can be seen in the values of the equilibrium constant, K, in Table 2. It is interesting to note that dissociated propylamine does not form the inclusion complex, while the dissociated propionic acid form the complex [14,15]. This means that the ionic group in guest molecules greatly influences the formation of the inclusion complex. It is found from the present study that the equilibrium constants for the systems with the dissociated forms of the acids are smaller than those for the systems with the undissociated forms. This result indicates that the cavity of β-CD can capture more effectively the undissociated guest molecules and the hydrophobic group.
Next, the results regarding the standard volume change of the reaction, ΔV, are considered. It was proposed that approximately five to seven water molecules are originally located inside the β-CD cavity in aqueous solutions and some of the molecules are expelled when a guest molecule enters the cavity. We simply determined that the standard volume change of the reaction is expressed as ΔV= nwVw – nCH2VCH2–VCH3, where nw indicates the number of expelled water molecules from the cavity of β-CD to the bulk, Vw is the molar volume of water, nCH2 is the number of methylene molecules, and VCH2 is the molar volume of the methylene group when butanoic acid are incorporated into the β-CD cavity [8]. The small standard volume changes of the reaction in this study indicate that the increase in the number of expelled water molecules compensates the increase in the incorporated hydrophobic group. Considering the molar volumes of water (Vw =18×10-6 m3 mol-1), the methylene group (VCH2 = 15.7×10-6 m3 mol-1), and the methyl group (VCH3=27.1×10-6 m3 mol-1) [13], the contribution of the incorporated methylene group to the volume change is smaller than that of the expelled water, although the values of nw and nCH2 are not definitely estimated. Then, with an increase in the carbon number of the guest, the volume change tends to decrease. Also, it can be considered that the ionized butanoic acid molecule is, to a certain degree, pulled out from the β-CD cavity compared to the nonionized butanoic acid. Therefore, the included portion of the butanoic acid molecule should be smaller than that under acidic conditions.
V. Conclusion
The observed ultrasonic relaxation in an aqueous solution, containing β-CD and butanoic acid, has been found to be due to the complexation reaction between β-CD and butanoic acid. The rate constants for the formation and the dissociation of the complex were obtained from the concentration dependence of the relaxation frequency and the maximum absorption per wavelength. The rate of the complex formation is found to be diffusion-controlled, taking into account the reaction site. Charge effect in the guest molecule is reflected in the dissociating process of the guest from the inclusion complex.