I. Introduction
II. Materials and method
2.1 Yeast strain
2.2 Ultrasound transmission system
2.3 Potassium Iodide (KI) dosimetry
2.4 Viability spotting assay
2.5 Heat shock assay
2.6 ROS (Reactive Oxygen Species)
2.7 ROS detection
III. Results
3.1 Cell death is induced by the optimized ultrasound condition
3.2 Heat shock effects on cell death
3.3 Reactive oxygen species on cell death
3.4 Biological mechanism of ultrasound induced cell death
IV. Conclusions
I. Introduction
Many applications of low frequency ultrasound from 20 to 200 kHz have been reported in physical, medical and biological domains. Low frequency ultrasound has used for diverse cleaning systems and medical therapeutic methods.[1] When blood vessels are clogged by thrombus or fibrin, 40 kHz ultrasound accelerates the thrombolysis or fibrinolysis.[2] Drug transport is enhanced by low frequency ultrasound, as it creates circulating eddies and acoustic pressure around oscillating bubbles that are formed by stable cavitation.[3] Ultrasound increases the membrane permeability and enhances the drug uptake of certain target cells such as HL-60 cells in vitro.[3] Low frequency ultrasound is also used for breaking kidney stones and for tearing fat tissue to assist liposuction.[1] It has been also reported that ultrasound irradiation with low frequency could be applied to induce chronic wound healing.[4]
For the studies of cell death, high frequency ultrasound over MHz has been widely applied to diverse cancer cells to induce cell death in cancer cells.[5,6] However, ultrasound is also used in monitoring of cell death images and could induce cell death.[7,8] Physical effects of low frequency ultrasound such as sonoporation of cell membranes are known as ways cells die under transmission. The biological effect of low frequency ultrasound via cavitation and ROS (Reactive Oxygen Species) formation is suggested as another way cell death occurs, but the mechanism is not fully understood. The precise mechanism and efficacious method of ultrasound induced-cell death remains to be elucidated.
Ultrasound at a frequency of 40 kHz is easily applicable and widely used in medical and biological devices. Therefore, we first developed the irradiation system by applying a 40 kHz ultrasound to the unicellular representative organism, yeast, Saccharomyces cerevisiae. The yeast cells are easy to manipulate and have many advantages including applica-bility to human cancer cells. Then, we examined the effect of 40 kHz ultrasound on the cell death and analyzed the thermal effect, radicals effects and other possible effects. We used the 50 W output of ultrasound generator and had worked in condition of standing wave. Therefore, the intensity and acoustic pressure are spatially not uniform and were not measured. In our study, we considered cell death dependent on the irradiated time instead of the intensity.
II. Materials and method
2.1 Yeast strain
The yeast Saccharomyces cerevisiae BY4743 was purchased from EUROSCARF (Germany). The obtained results are applicable to human cells. The strain was incubated in 5 ml YPD medium (50 g/l, GIBCO, BRL) at 25 °C shaking incubator of 200 r/min.
2.2 Ultrasound transmission system
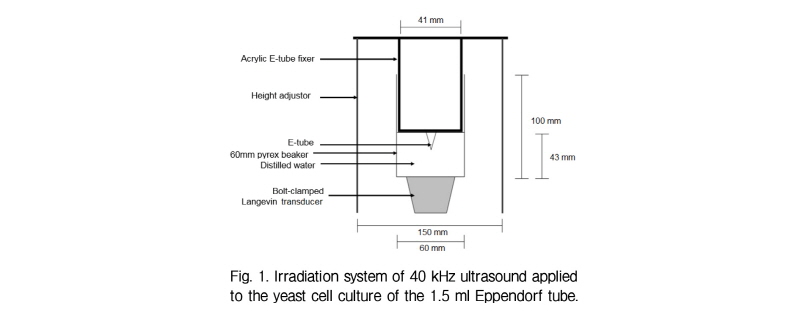
A pyrex beaker of outer diameter 60 mm was used as an ultrasonic transmission cell. A BLT (Bolt-clamped Langevin Transducer) with a 40 kHz nominal frequency was attached at the bottom of the transmission cell. The BLT was excited by an ultrasonic generator (50 W Power, Kodo Technical Research Co., Korea). Two vertical height adjustors were placed 15 cm apart. At the middle of the two adjustors, the transmission cell was fixed using a bolt. Distilled water was poured into the beaker, and ultrasound was transmitted using an ultrasound producer.
In order to transmit a 40 kHz ultrasound into the yeast cell culture of the 1.5 ml Eppendorf tube, a stable Eppendorf tube fixing system was devised using acrylic. (Fig. 1) A U-shaped acrylic extension was added to the middle of 160 mm × 38 mm acrylic. In the middle of the U-shaped extension, a hole of 10 mm was made to fix the Eppendorf tube.
2.3 Potassium Iodide (KI) dosimetry
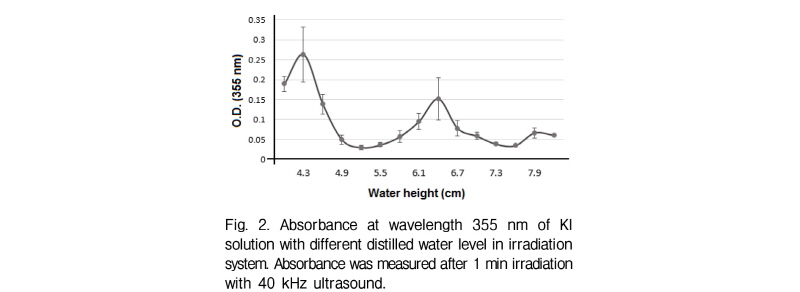
The OH radicals attack Potassium Iodide (KI) and liberate iodine. When liberated iodine accumulates, I3- forms in the solution. I3- of ultrasound irradiation at certain level of water surface was estimated by measuring the absorbance of KI solution at 355 nm wavelength.
2.4 Viability spotting assay
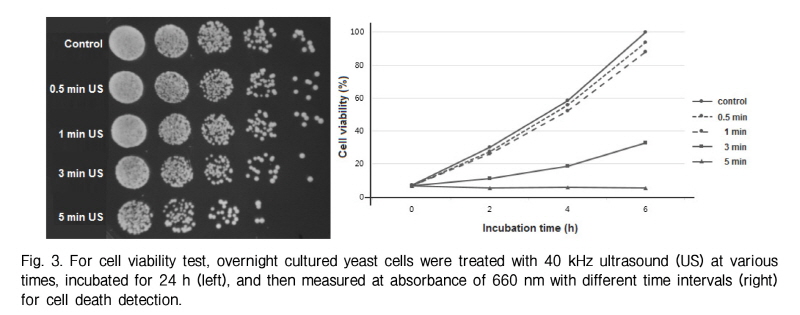
The yeast cell culture was prepared and adjusted with optical density 0.1 at 660 nm wavelength (OD660 = 0.1). The solution was diluted one fifth for four times to prepare solutions with concentration of 1/5, 1/25, 1/125, 1/625 of the original solution. 10 ㎕of series of diluted solutions are spotted on the YPD-agar plate (15 g/l) in a row, and incubated for 24 h before colony was counted.
2.5 Heat shock assay
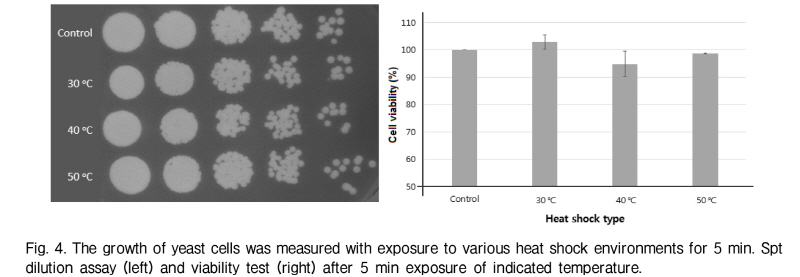
The yeast cell was plated on YPD-agar media (15 g/l) and incubated with 5 min at normal condition at 25 °C. It was also incubated for 5 min under heat shock conditions at 30 °C, 40 °C, 50 °C in a waterbath. Temperature was controlled within ± 1 °C. After heat shock, the yeast cells were returned to the normal condition and incubated overnight.
2.6 ROS (Reactive Oxygen Species)
Hydrogen peroxide (Sigma-Aldrich, USA) of 0.1 mM, 0.5 mM and 1 mM were treated to each yeast cell culture (O.D.660 = 0.1) to induced condition of Reactive Oxygen Species. ROS was inhibited by adding different concentrations of NAC (N-acetyl-L-cysteine; ENZO Life Sciences) to cells right before ultrasound transmission.
2.7 ROS detection
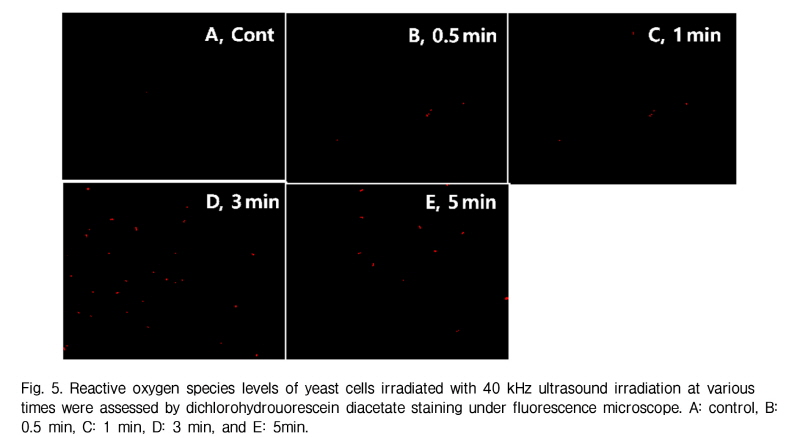
ROS formed due to ultrasound irradition can be viewed under a fluorescence microscope when stained with ROS reaction dyes. Oxygen radicals were induced by adding pyocyanin to negative control cell. ROS was detected by adding 10 g/ml 2`,7'-dichlorohydrouorescein diacetate (Sigma- Aldrich, USA) to yeast cell cultures (O.D.660 ≈ 0.4). Cells were left to grow for an additional two h and then viewed under Zeiss Axiovert 40 Fluorescence microscope (Zeiss, Swiss).
III. Results
3.1 Cell death is induced by the optimized ultrasound condition
An irradiation system with ultrasound at 40 kHz frequency was developed (Fig. 1) and used to test the KI ionization for optimal position. Fig. 2 shows the optical density values of KI solution at 355 nm after 1 min of 40 kHz ultrasound transmission at different water heights. The two peaks at 4.3 cm and 6.4 cm mark the water height at which oxygen radicals formed the most.[9] This indicates that at these water height (4.3 cm and 6.4 cm), the 40 kHz ultrasound is successfully transmitted to the Eppendorf tube placed at the water surface level. Another point worth noting is that the distance between two antinodes (2 cm) is similar to the theoretically calculated distance between antinodes of 40 kHz ultrasound in water. Among the two peaks, 4.3 cm water height was chosen as the optimal condition for 40 kHz ultrasound transmission, since absor-bance is higher than that of 6.4 cm.
The cell viability of yeast exposed to 40 kHz ultrasound for different durations was detected in Fig. 3. Cell death is induced from 3 min to 5 min of ultrasound transmission. This result implies that our optimized ultrasound irradiation system successfully transmits ultrasound to yeast cells (diameter 5∼10 µm) and induces cell death.
3.2 Heat shock effects on cell death
During the ultrasound transmission, hyperthermia occurred under the tested conditions. The temperature of the system increased a maximum temperature of 50 °C when the system was irradiated by the 40 kHz ultrasound for 5 min. Since hyperthermia has an important role in ultrasound effects, we checked whether exposing yeast cells to high temperatures for 5 min would induce cell death or not. In this condition, the experiment failed to show that heat shock induced cell death (Fig. 4) This result suggests that hyperthermia of the irradiation system is not relevant to 40 kHz ultrasound induced cell death.
3.3 Reactive oxygen species on cell death
It is well known that ultrasound stimulates inertial cavitation and oxygen radials. ROS accumulated in the yeast cells irradiated with 40kHz ultrasound for 3 min and 5 min (Fig. 5). This results correspond with that of cell viability for 3 min and 5 min. Thus, the result suggests that reactive oxygen species may be involved in 40 kHz ultrasound using this transmission system.
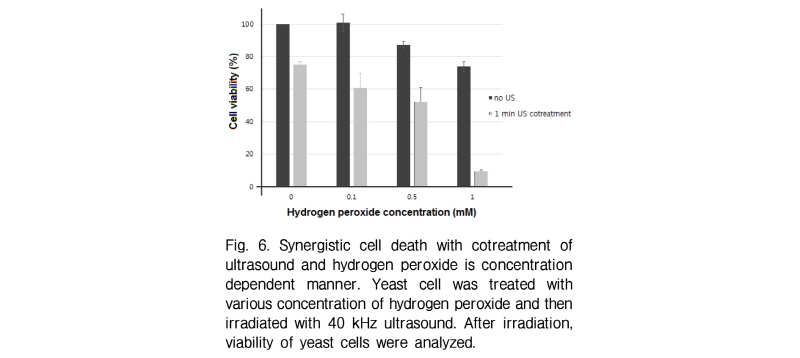
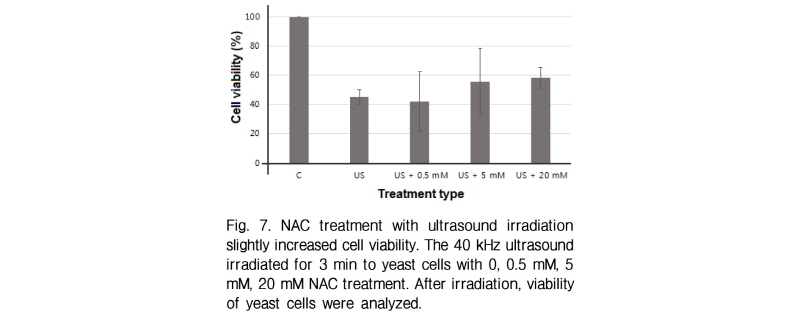
If ultrasound induced cell death is closely related to reactive oxygen species formation, extraneous oxygen radicals will be promoted by the effect of ultrasound transmission. To check this, yeast cells were co-treated with hydrogen peroxide and ultrasound irradiation. Interes-tingly cell death is synergistically induced in ultrasound irradiated cells with hydrogen peroxides (Fig. 6). It must be noted that the amount of cell death induced by co-treat-ment is larger than the sum of cell death induced by two treatments independently. If ultrasound is one of the reactive oxygen sources which can trigger cell death, the inhibition of reactive oxygen species will suppress the cell death by ultrasound. During 3-min ultrasound irradiation test, yeast cells were treated with different concentrations of NAC as a well known reactive oxygen scavenger. NAC treatment with yeast cells slightly inhibited cell death induced by 40 kHz ultrasound (Fig. 7). These results suggest another way of cell death induced by ultrasound beyond that of reactive oxygen species generated by ultrasound.
3.4 Biological mechanism of ultrasound induced cell death
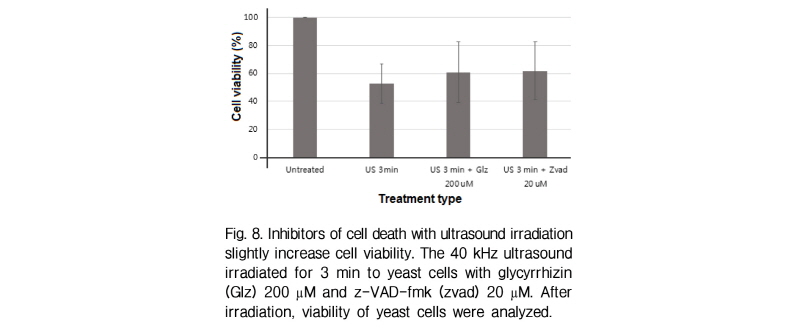
The biological patterns of cell death are necrosis and apoptosis which can be induced by various stress agents including physical and chemical stresses.[10,11] When yeast cells containing glycyrrhizin (necrosis inhibitor) or z-vad-fmk (apoptosis inhibitor) are treated with 40 kHz ultrasound, cell viability is slightly increased (Fig. 8). This result suggests that either necrosis and apoptosis are not major types of cell death or chemical reagents cannot inhibit both types of cell death in this transmission system.
IV. Conclusions
In this study, a system that transmits 40 kHz ultrasound was developed and optimized to study yeast as a representative unicellular organism. Using an ultrasound transmission system, yeast cell death was examined. We found that cell death is dependent with irradiation time. We used the 50 W output of ultrasound generator and had worked in condition of standing wave. Therefore, the intensity and acoustic pressure are spatially not uniform and were not measured. In our study, we considered cell death dependent on the irradiated time instead of the intensity. Large amount of cell death was observed after 3 min of ultrasound irradiation.
After the optimization of the ultrasound transmission system, a mechanism of cell death was examined. The thermal effect of low frequency ultrasound was not plausible to ultrasound induced cell death because the viability of heat shocked cells was similar to that of control cells (Fig. 4). A reactive oxygen species was suggested as one of the cause of cell death induced by ultrasound, as hydrogen peroxide and ultrasound showed synergistic effects in inducing cell death. Also, the amount of reactive oxygen species formed corresponded with the amount of cell death occurred as ultrasound transmission time increased. Therefore, it was assumed that ultrasound induced yeast cell death might be involved in reactive oxygen species generation. However, the treatment of NAC did inhibit slightly the ultrasound induced cell death. Necrotic and apoptotic cell death were also slightly inhibited by each specific cell death inhibitor. Taken together, this study suggests that ultrasound induced cell death may be not exclusively related to reactive oxygen species formation in the cell and that other cell death pathways other than apoptosis and necrosis must be carried out.
It shoud be noted that applying these results to cell deaths studies may be difficult because of non-linear effects of low frequency ultrasound.