I. Introduction
II. Vibration effect
2.1 Vibration profiles of vehicles
2.2 Electrochemical performances by vibration
III. Shock and penetration effect
3.1 Mechanical shock on batteries
3.2 Penetration on batteries
IV. Ultrasound effect
4.1 Relation of ultrasound to electrochemical reaction
4.2 Exploitation of ultrasound to batteries
V. Perspective
I. Introduction
As industrialization progresses around the world and the use of fossil-fuel increases, the seriousness of global warming is arising.[1,2] To break through the issue, many companies and governments have been trying to overcome threats in many ways. In the field of the car industry, they are actively participating in the movements, transiting power supply system from Internal Combustion Engines (ICE) to Lithium-Ion Batteries (LIBs).[3,4] To make the successful process of changing, it is required for the vehicle industry to include features coming from the differences between Electric Vehicles (EVs) and conventional ICE-based vehicles. For instance, vehicles are operated under various environmental conditions. (e.g., vibration, shock). However, LIBs have been developed and used for static systems. Accordingly, it is demanded EVs to consider LIBs characteristics,[5] or redevelop LIBs with considerations of dynamic systems.
In a view of LIB redesign, just a few studies about vibration and shock effects on LIBs have been done so far and the implant of battery systems on vehicles may not work effectively.[6,7,8,9] It is known that external forces such as vibration/shock can seriously deteriorate the electrochemical performance of batteries.[10,11] To prevent the gradual performance degradation, researchers have focused on the analysis of the relationship between the battery and the force.[12] For instance, by applying random vibration profiles or a specific frequency, it has been found that vibration can cause performance degradation of the battery.[13,14,15,16] When powerful shocks were applied to batteries, LIBs displayed life time end by sudden failure or structural destruction.[15,17]Meanwhile, electric vehicles are typically situated in vibration consisting of a wide range of frequencies.[18,19,20,21] Though the high frequency (i.e., ultrasound) takes a small portion of the whole vibration, ultrasound has a great influence on the electrochemical performance of LIBs, altering the electrochemical behavior inside LIB systems.[22,23,24]
Here we discuss the comprehensive relationship between LIBs for the first time. In the first section, we explain the effect of dynamic forces on batteries for electric vehicles. (e.g., normal vibration, shock, and ultrasound) In section 2, vibration test profiles and the effect of vibration on batteries are introduced. Then, it is described how mechanical shock, including penetration, affects LIBs in section 3. Section 4 presents the relationship between ultrasound and several batteries. Last, a summary with perspective is discussed. The table of contents is shown below.
1. Introduction
2. Vibration effect
2.1. Vibration profiles of vehicles
2.2. Electrochemical performances by vibration
3. Shock and penetration effect
3.1 Mechanical shock on batteries
3.2 Penetration on batteries
4. Ultrasound effect
4.1 Relation of ultrasound to electrochemical reaction
4.2 Exploitation of ultrasound to batteries
5. Perspective
II. Vibration effect
2.1 Vibration profiles of vehicles
As cars with ICEs become popular, the issue of car safety has always been considered seriously. Since cars are consistently exposed to vibration, researchers have analyzed the effect of vibration from standard driving tests for vehicles with internal combustion engines.[25,26,27,28] However, due to various vibration types by different driving conditions and complex components in vehicles, it is still lack of perfect examples for vehicle vibration profiles.[21,26,27] Though, Wang et al.[26] designed a reliable sinusoidal vibration profile by using variable amplitude. They chose vibration amplitude from filtered time series field data instead of order analysis results, which underestimated the vibration amplitude.[26] Ohta et al.[27] considered the effect of vibration from piston movement while driving. They calculated the effect of piston slap by analyzing the time step integration technique showing different force distribution with the change of frequency [Fig. 1(a)].
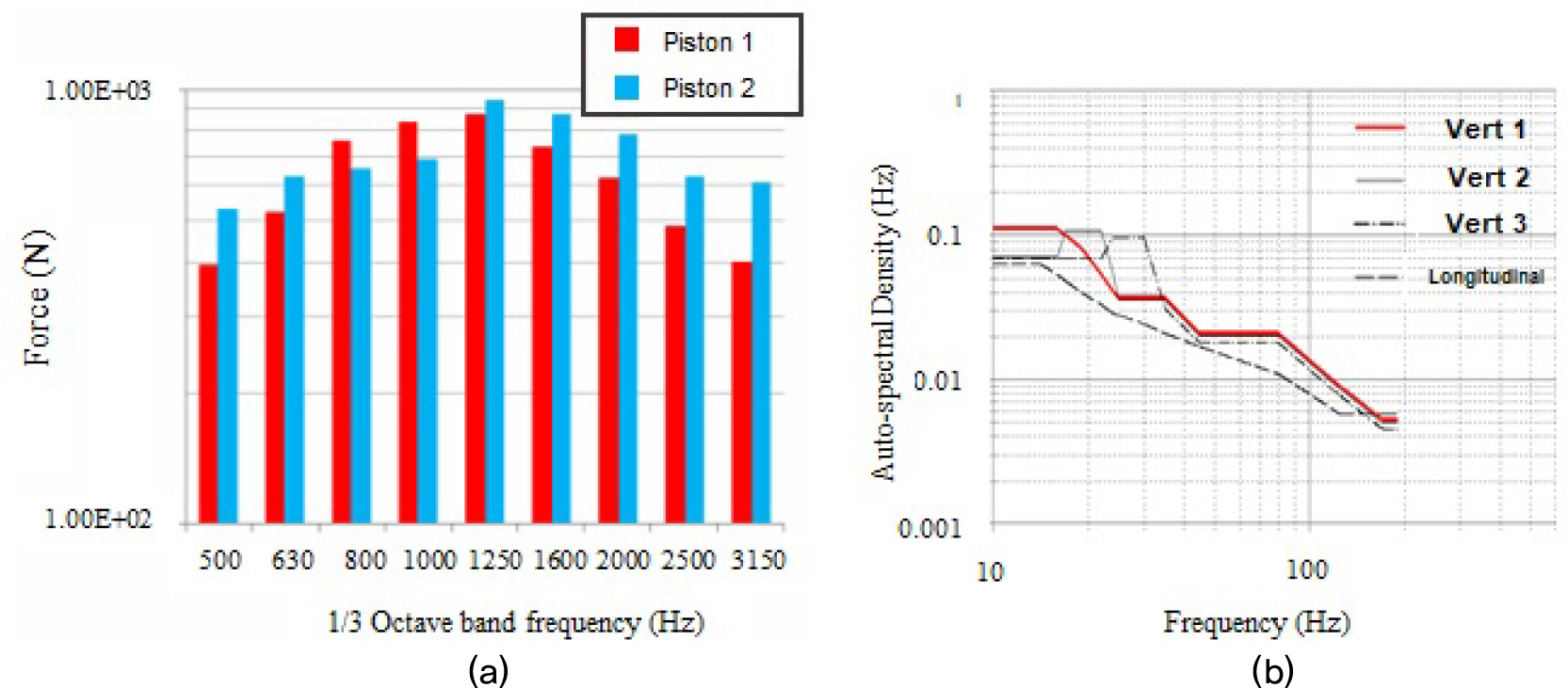
Fig. 1.
(Color available online) Measured vibration profile from ICEs and EVs. (a) The frequency spectrum of pistons in an internal combustion engine. Modified from Reference [27] with the copyright of JSME. (b) Auto-spectral density vibration profile from SAE J2464. Modified from Reference [32] with the copyright of Lockheed Idaho Technologies Company.
While transiting ICEs to EVs, it is limited in directly applying the previous evaluation criteria to EVs.[9,29] For example, Lang and Kjell showed differences between gasoline-based and electric-based vehicles with Power Spectral Density (PSD).[30] Meanwhile, in the early 2000s, it began to look for vibration profiles suitable for EVs. For instance, SAE J2464 is one of the earliest tests for batteries applied in electric vehicles.[9] The profile includes not only vibration and temperature tests, but also external short circuit tests. United Nations (UN) also published EV- aimed testing protocol UN 38.3 T3 which is currently considered a standard vibration profile, especially for product transportation.[31] United States Advanced Battery Consortium LLC(USABC) compiled the cumulative shock pulses near locations relevant to batteries so as to get a profile over the life of vehicles up to 100,000 miles.[32] In detail, Auto-Spectral Density (ASD) of vibration is shown in the frequency range of 10 Hz ~ 190 Hz with vertical, longitudinal directions, which includes random vibrations with 0.1 g2/Hz [Fig. 1(b)]. It is worthy to mention that the ASD profile was not proper for real-time vibration test, for it without mirroring real-operation of vehicles.[31] European Union (EU) also reported a similar vibration profile called ECE Regulation 100. It well copies the normal driving of the vehicle with a frequency range of up to 50 Hz. However, similar to previous vibration profiles, it still remained a short-term test of batteries.[31,32] Moreover, ECE Regulation 100 only considers a vertical axis, not X or Y mounting axis for batteries.[33]
Considering differences between standard vibration profiles and actual driving conditions, vibration profiles need to be rebuilt based on real data. Hooper and Macro[20] compiled vibration profiles from urban, rural and motorway surface conditions with different models. They used Power Spectral Density (PSD), which represents that random vibration is imposed on their test. The authors argued that it is important to differentiate the test standard regarding the sort of electric vehicles with the vibration profile in a city road condition. Unlike previous vibration profiles focusing on frequencies higher than 10 Hz,[32] Hooper and Macro’s vibration profiles include low frequency range below 7 Hz.[31] Also, the authors built a PSD plot for 100,000 miles of vehicle life which showed a loading peak between 20 Hz and 40 Hz caused by loads through the structural element. Especially for iMiEV, PSD showed much lower vibration loading compared with SAE J2380. The reason is that SAE J2380 doesn’t represent typical driving of EVs.[18,31]
2.2 Electrochemical performances by vibration
With the development of vibration profiles based on real-driving conditions, collected profiles were applied to investigate the effect of vibration on batteries in electric vehicles.[13,14,15,34,36,37] Brand et al.[15] imposed UN 38.3 T3 on lithium-ion pouch cells and lithium-ion 18650 cylindrical cells. In the pouch cell, internal resistance gradually increased, and discharge capacity decreased with every direction. On contrary, cylindrical cells were largely affected by z direction and CT showed the shape change of mandrel. Hooper et al.[36] investigated the effect of vibration on Nickel-Manganese-Cobalt (NMC) 18650 cells. The authors applied two vibration profiles (SAE J2380 and WMG/Millbrook) at various State Of Charges (SOCs) and with various vibration axis. The result indicates vibration increased cells’ discharge capacities and internal resistances regardless of vibrating directions.
To analyze the increase of internal resistances, the authors investigated the surface chemistry of NMC 18650 cells using X-ray Photoelectron Spectroscopy (XPS).[35,36] Vibration damaged surface of electrodes’ and a relatively high resistance, (e.g., O – C = O) replaced the broken site. It is worth mentioning that the resistive component mostly exists on the electrode surface (Fig. 2).[38] Meanwhile, unlike that a range of random vibrations was applied on batteries,[34,35,36] Zhang et al.[13] correlate battery performance with mechanical vibration at specific frequencies. They insisted that vibration increases the internal resistance of batteries, especially in the frequency range lower than 30 Hz. Shelke et al.[37] also reported that vibration can increase LiNixMnzCoyO2 (NMC), LiFePO4 (LFP), and LiCoO2 (LCO) cells’ internal resistance, implying that vibration can induce performance degradation regardless of cathode’s material. Parasumanna et al.[16] further confirmed the findings with SEM, showing that the increased internal resistance is caused by the breakage of electrode surfaces. Considering the increase of internal resistance can block charge transfer on the electrode surface, it may be required for the battery package to block the transfer of vibration inside.
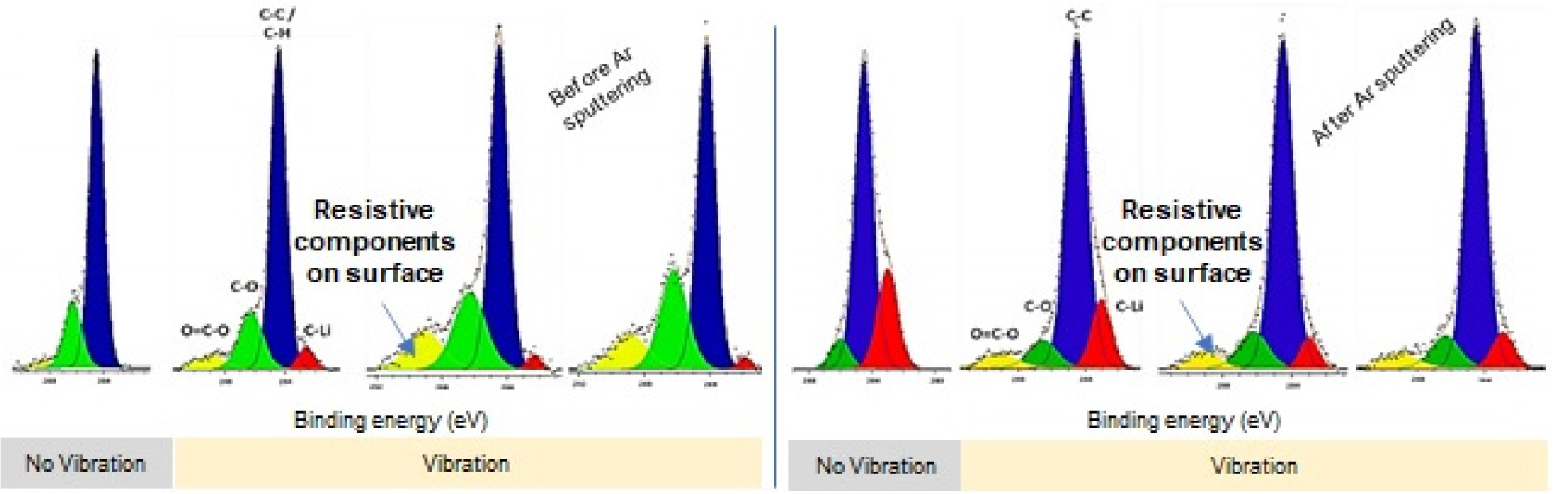
Fig. 2.
(Color available online) Surface chemistry of cells with and without vibration. Modified from Reference [35] with the copyright of MDPI.
III. Shock and penetration effect
3.1 Mechanical shock on batteries
Batteries can be exposed to impact load as well as cyclic load during their application. For example, Electric car driving on a road suffer vibration load especially when the road has rough surface. It is also possible that foreign objects with high-speed attack battery embedded in the car. The sudden impact on batteries leads to catastrophic outcome. Therefore, it has been requested to understand the mechanical response causing thermal and electrical failure.[14,15,19,25] Accordingly, it needs to be well examined about the response of batteries to extreme loads which increases the risk of structural failure and short circuits.[39,40,41,42] Tobishima et al.[43] carried out the crash test using commercial cylindrical 18650 cells. The authors investigated temperature change of cells by short circuit hit for fully charged cells stored at varied temperatures and periods. They found that the failure behavior of a cell is independent to age and temperature. Kim et al.[44] simulated the deformation of cylindrical cells by dynamic load using the Finite Element Method (FEM) with a 3D homogenized model. Afterward, Gilaki and Avdeev exploited the 3D homogenized material model for cells at a high-strain rate.[45] Then, they analyzed failure scenarios with different impacts. In addition, they conducted a drop- weight experiment and FEM analysis.[46] Though homogenized models allow to predict mechanical deformation of cells by impact,[45] it has limitation to predict the large distortion of each component of a cell in detail. In this regard, Gilaki and Avdeev applied heterogeneous material models on cells. That is, different material properties were used for anode, cathode, and separator.[47] The authors argued that separator play an important role in the overall abuse tolerance. They found that short circuit occurs first in separator due to poor mechanical strength of a separator.[47] Hu et al.[48] investigated the effect of impact velocity on LIBs with a variety of SOCs and showed the dependency of maximum stress on SOC. The tendency was observed in all the test of bending, radial compression, and indentation in a similar way. However, it is known that cells with different SOC have different thermal runaway temperature.[49] That is, trading-off characteristics of batteries in terms of SOC should be taken into account. Meanwhile, when batteries are abused by an unknown object, the shape of intruding body is unpredictable. Sahraei et al.[50] indent pouch cells with various punching tools. Considering the batteries being subjected to dynamic load, countless failure scenarios may be available. Although the previous studies revealed some effects of dynamic load on batteries, they have the following limitations. First, the poor reproducibility of the test makes their reliability suspicious. Also, experiments have difficulty in covering all the scenarios of accidents. Therefore, further studies examining underlying mechanisms of battery during sudden shock are crucial to use battery safely.
3.2 Penetration on batteries
Penetration is an invasive mechanical way to produce local deformation and fracture causing an internal short circuit. Therefore, simulating penetration is considered crucial to investigate safety in a cell. Despite the importance, only a few studies were reproducible and properly represents the actual failure situation.[40,52,53,54,55,56] The reason is that penetration tests can be affected by various parameters, such as SOCs, nail size, speed, location of penetration, etc.[57] Tobishima and Yamaki[51] evaluated the effect of nail penetration on prismatic cells. They compared normal and overcharged cells. Unlike the usual cells, overcharged cells smoked with nail penetration, while cells charged at the standard did not. From the result, they confirmed that cell safety is sensitive to state of charge and thus argued that charge voltage should be precisely controlled. Similar studies further confirmed Tobishima and Yamaki’s observation.[54,58,59] Meanwhile, in real applications, it is difficult to predict where intrusion occurs in a battery. For that reason, penetration data should be collected on diverse positions of a cell. Maleki and Howard[53] systematically conducted the nail penetration at 6 different locations. They observed that the temperature near edge part is higher than center area because of different heat dissipation on edge and center. Therefore, when designing batteries, the difference of potential to thermal runaway could be regarded as a designing factor that affects the safety of batteries. Although several abusive tests have successfully provided insight about cell design in a view of cell safety, only a few reports describe what happens inside a battery during penetration. Wang et al.[60] theoretically built a scenario of ion flow after short circuit by penetration (Fig. 3). In real applications, a pointed body can penetrate a cell at various speeds, and accordingly the intrusion velocity effect must be also covered. Santhanagopalan et al.[61] investigated the mechanical behavior of a cell at varied intrusion speed up to 1000 m/s. The most significant problem in intrusion test of a cell is tremendous heat generation. For that reason, many studies systemically analyze the heat generation and temperature distribution inside batteries.[60,61,62,63,64,65,66] So far, many researches have examined the failure scenarios of penetration on battery. However, the perfect solution for these scenarios has not been found. Application of high thermal stability materials or decelerating the active materials’ thermal reactions would be conceivable strategies to save batteries from thermal runaway during nail penetration.
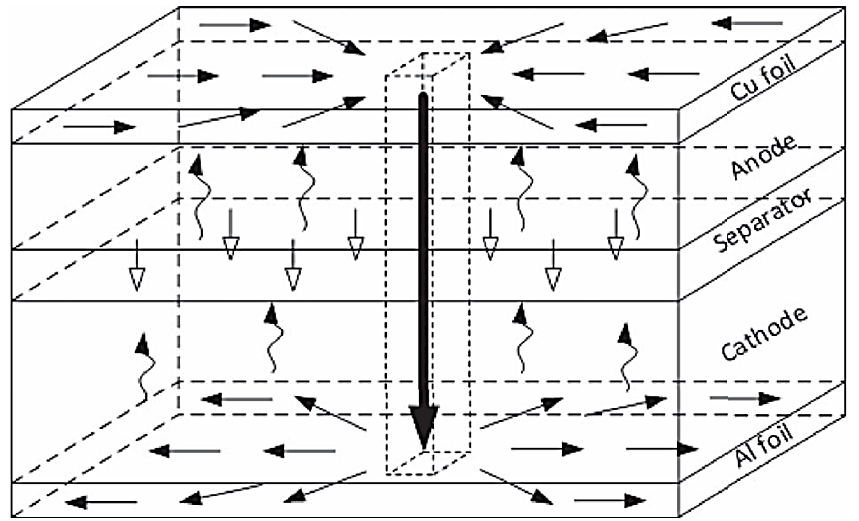
Fig. 3.
Schematic of electrons and ions in a cell during penetration process. Modified from Reference [60] with copyright of IOP publishing limited.
IV. Ultrasound effect
4.1 Relation of ultrasound to electrochemical reaction
Ultrasounds with low- and high-intensity were used to non-destructive test and cleaning so far, respectively.[67,68,69,70] With the recent rise of batteries, science that studies the effect of ultrasound on chemistry has drawn great attention, showing noticeable progress.[71,72,73,74,75,76,77] Chatakondu et al.[73] insisted that ultrasound enhances intercalation, perhaps indicating increase of reaction rate and capacity. They found that organic and inorganic molecules intercalated well into layered solid hosts at 20 kHz ultrasound.[73] It is worthy to mention that the authors applied high acoustic intensity (ca. 20 W/cm3) on host materials and, thus bulk host was pulverized into small pieces. That is, the enhanced intercalation may be originated from the reduced diffusion (i.e., size effect),[78] not from improved kinetics. Due to the destructive feature of acoustic waves to material, ultrasound can help to plate ions on the electrode in a flat shape and accordingly suppress dendrite formation in metal plating.[76] For instance, Zhou et al.[72] made a flat Ni electrode surface using two directions of ultrasound.
In moderate ultrasound intensity, the effect of ultrasound on kinetics would be more apparent rather than the size effect. For instance, Yuwen et al.[79] found that n-butyl Li readily intercalated into layered transition metal dichalcogenide sheets with the aid of ultrasound. They attributed the enhanced kinetics to the generation of shockwave by acoustic cavitation which induces turbulence of solution and thus increases mass transfer.[79]
4.2 Exploitation of ultrasound to batteries
With lots of studies about the effect of ultrasound to electrochemical reactions, researchers focused on the correlation between LIBs and ultrasound. Ding et al.[80] studied how lithium metal anode reacts to ultrasound in high charging rates, which was an early study of ultrasound and next-generation batteries. Ultrasound could effectively lower overpotential and internal resistance at the surface of metal anodes, referring to enhanced electrochemical performance. Friend et al.[81] applied Surface Acoustic-Wave (SAW) on lithium metal batteries and showed a flattened lithium surface. They simulated how ultrasound homogenizes lithium-ion concentration and how subsequent uniform current flux affects dendrite formation (Fig. 4). They also observed that the dead lithium layer is formed less and thus lift time increases. Furthermore, the relationship between dendrite shape on lithium metal surface and ultrasound power was analyzed through microstructure images. The authors claimed that ultrasound makes uniform lithium-ion concentration. However, if ultrasound is too powerful, it leads to the formation of a thick dead lithium layer.[22] SEM images represent how the shape of dendrite behaves differently according to the power of ultrasound, which is named Enhanced Mass Transfer (EMT). Recently, Tang et al.[23] found that even in the lithium-oxygen battery, ultrasound can have positive effects. The authors argued that ultrasound not only enhances mass transfer but also improves the reaction kinetics of oxygen. The process with ultrasound makes Co3O4 react active on the electrode’s surface, indicating that ultrasonic could be a source of additional energy. These studies imply that acoustics can be utilized for energy storage systems in versatile ways.
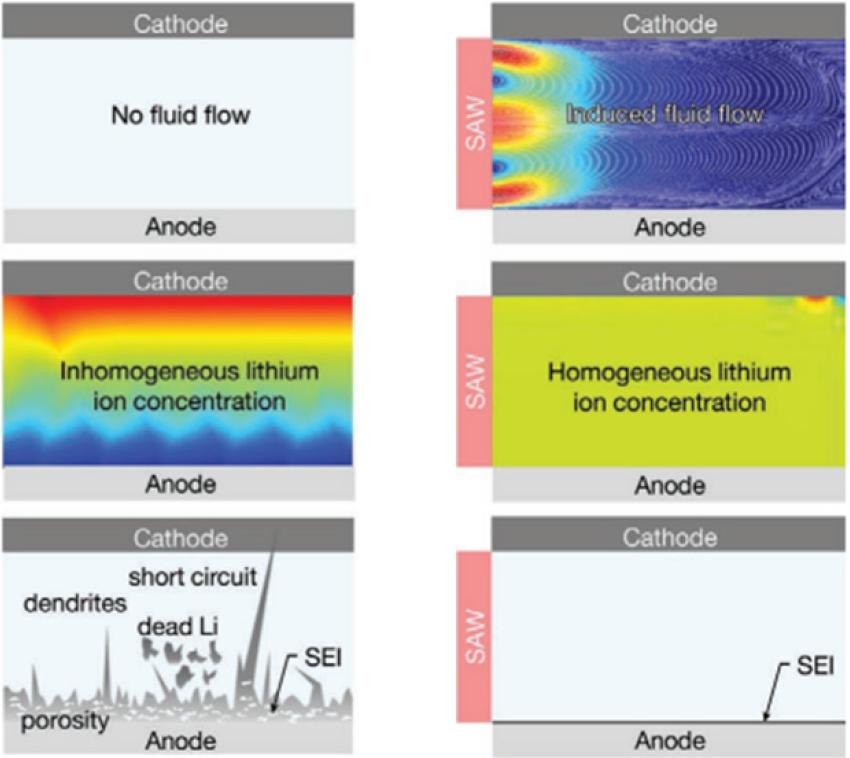
Fig. 4.
(color available online) comparison of how SAW affect lithium-ion concentration. modified from Reference [81] with copyright of wiley.
V. Perspective
EVs are exposed to various environmental conditions such as normal vibration, shock, ultrasound, and thus their power supply systems, battery modules, undergo similar dynamics. However, batteries have been developed based on static applications. Therefore, it should be needed for LIBs to consider dynamic variables (e.g., vibration and shock) in their design. As discussed in section 2, mechanical vibration reduces capacity. Therefore, more LIBs should be mounted on EVs than initial energy calculations. Also, it must be aware that secondary components of batteries such as mandrel are often destroyed even by vibration, and it leads to cell failure. With this in mind, structural components of LIBs for EVs may need more strict mechanical properties. In a view of LIB- housing design, cell cases must be designed on the basis of penetration at the edge area, not the center.
Meanwhile, isolating the vibration from the vehicle chassis to the battery pack can be the fundamental solution to resolve the issues. Vehicles are exposed to various vibration inputs, which create a wide range of excitation frequencies. The vibration frequencies transmitted to batteries should be suppressed to avoid performance degradation or the resonance of the battery pack structure. Conventionally, assembled battery cells are not thoughtfully designed to isolate the transmission of undesirable vibrations. Thus, the development of technologies in vibration isolation structures must be provided to achieve stabilization of structure within the battery pack enclosure.
As discussed in section 2.2, vibration deteriorates batteries’ structural stability and solid electrolyte interphase (SEI). However, ultrasound enhances electrochemical performances with enhanced surface kinetics, resulting in low charge transfer resistance. Unlike batteries, SEI does not exist on fuel cells and electron transfer occurs on the interface between electrode and electrolytes. Therefore, it is likely that electrochemical reactions of fuel cells can be boosted by the aid of ultrasound. On the other hand, mechanical vibration may have a negligible effect on them. In this regard, the knowledge obtained from batteries could be implanted into fuel cells. Tough, the two systems depend on different electrochemical mechanisms.
This paper illustrates problems for batteries in EVs in both views of the battery pack and EV chassis. Overall, mechanical vibration, including shock, on batteries can deteriorate cells’ electrochemical performance. As discussed, to avoid the issue, one choice may be forced to us between the redesign of the battery or the body frame. However, developing LIBs for dynamic systems, or redesigning packs isolated to vibration would not work completely. For this reason, to overcome effects of dynamic loads, solutions must be discovered integrating vibration-resistive batteries and vibration-isolated structures.




